Everything around us is made of matter, and it can exist in many different forms. In this post, Laura explains the main states of matter and explores some extraordinary ones beyond the everyday.
State of matter is commonly defined as “one of the distinct forms in which matter can exist” [1], where each state is distinguished by how the particles are arranged and how they behave collectively. Most people agree that in our day-to-day interactions, there are four states of matter: solid, liquid, gas and plasma.
What are the differences between each state?
In a solid, the forces of attraction between particles are very strong, resulting in particles vibrating about fixed positions and the solid having a definite volume and shape. In a liquid, particles have a higher amount of kinetic energy, which causes them to overcome the force of attraction between particles slightly. This allows the particles, even though they are still close together, to be able to move more freely. Hence, liquids take the shape of their container (no definite shape) but have a definite volume. A gas is a compressible fluid where the particles have so much kinetic energy that they overcome the forces of attraction between them completely. This means gases have no definite shape or volume and fill the container that they are in. Finally, we have plasma which is an ionised gas. Gases can be converted to plasma in one of two ways: by adding electricity or heating them to an extremely high temperature. This causes some of the particles to lose electrons and gain a positive charge, and in turn, these positive charged ions are surrounded by the electrons that were lost. This is why plasma can conduct electricity. Plasma is present in neon lights and lightning, and it makes up almost 99% of ordinary matter in the universe, as it forms all stars [1].
The Bose-Einstein Condensate
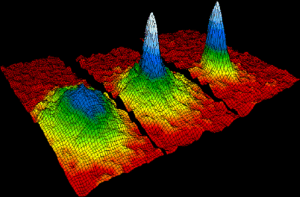
There are also many non-classical states such as the Bose-Einstein Condensate (BEC), which some consider to be the fifth state of matter. This state occurs when separate atoms or subatomic particles are cooled to absolute zero (0 K or -273.15 °C). This was predicted by Albert Einstein and Satyendra Nath Bose in 1924 [5].
To better understand this concept, we can zoom in on a subatomic scale. According to quantum mechanics, subatomic particles can be considered as small wave packets – “short bursts of localized wave action that travel as a unit, formed by combining multiple waves of different frequencies” [8]. When particles cool down to near absolute zero, their wavelengths expand causing them to overlap with each other. The particles then become a single quantum entity with the same wave function and stop behaving as individual particles [4]. If you were to observe them, you would see a blur instead of individual particles.
Some materials which contain BECs include superconductors, which conduct electricity without any resistance when cooled below a certain threshold [7], and superfluids, which are fluids that flow without viscosity and lose no energy as they move. This only occurs at extremely low temperatures [6]. Some uses of BECs are in quantum computing, researching dark matter and other scientific research.
Production of Bose-Einstein Condensates
Although BECs were predicted in 1924, they were not actually produced until 1995 because the necessary technology way not yet available. They were produced by Eric Cornell and Carl Wieman who made BECs with Rubidium atoms, and by Wolfgang Ketterle who also did so in the same year using sodium atoms. All three scientists received the Nobel Prize in Physics in 2001.
But how do we actually achieve such a low temperature? Firstly, free atoms of the same element are hit with lasers from all three axes (six directions in total). You may be wondering how lasers cool something down – the laser light hits the atoms, causing them to recoil, which slows them down, hence their temperature also goes down. A magnetic field is used to trap the atoms and remove any hotter atoms until the desired temperature is achieved. However, BECs are very fragile, meaning even a slight change in the environment can be enough to warm them past the condensation threshold [4].
Reference List
- Wikipedia. State of matter (2025). Retrieved from https://en.wikipedia.org/wiki/State_of_matter#
- The organic Chemistry Tutor (2019). States of Matter – Solids, Liquids, Gases & Plasma – Chemistry. Retrieved from https://www.youtube.com/watch?v=9TVOlTolKFA&forced
- Science ABC (2023). Bose Einstein Condensate Explained in Simple Words. Retrieved from https://youtu.be/KJU4vZMHegg
- Ninergy (2019). 5th state of matter Bose-Einstein Condensate. Retrieved from https://youtu.be/Vv_ED4F1tAk
- Sidney Perkowitz (2025), Bose-Einstein condensate. Retrieved from https://www.britannica.com/science/Bose-Einstein-condensate
- Wikipedia. Superfluidity (2025). Retrieved from https://en.wikipedia.org/wiki/Superfluidity
- Wikipedia. Superconductivity (2025). Retrieved from https://en.wikipedia.org/wiki/Superconductivity
- Wikipedia. Wave packet (2025). Retrieved from https://en.wikipedia.org/wiki/Wave_packet